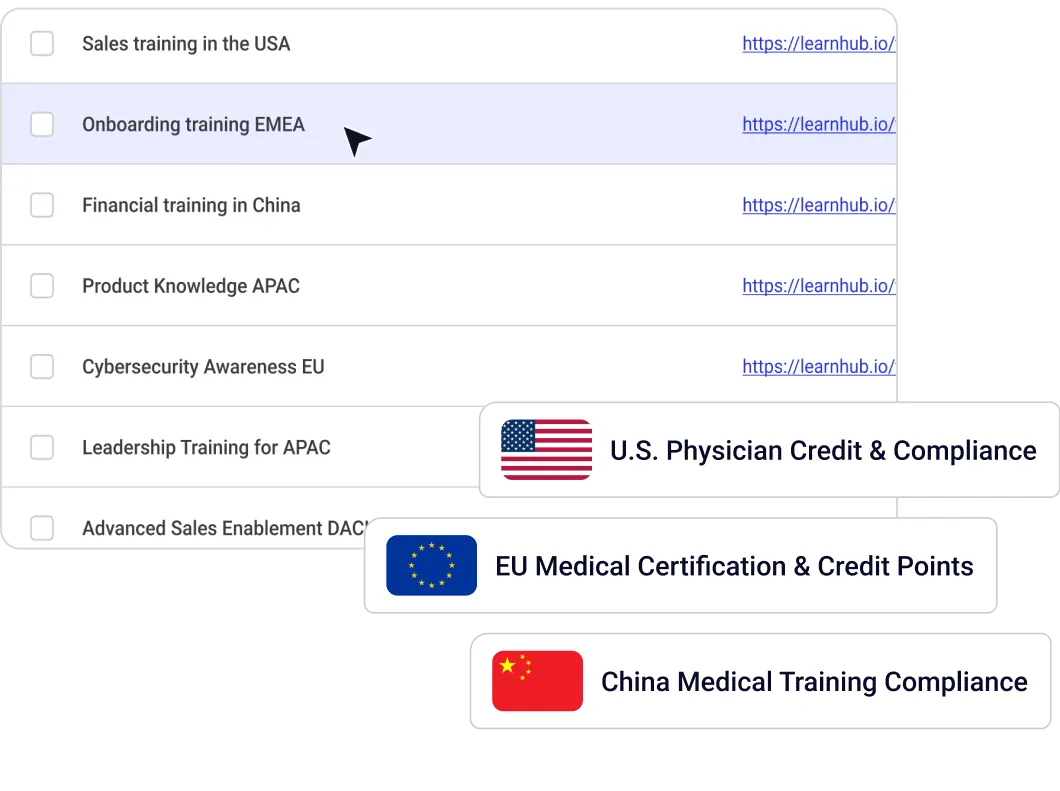
Pharmaceutical and healthcare organizations face a training challenge that most LMS platforms were not designed for: delivering compliant, validated, and continuously updated training simultaneously to multiple distinct audiences — internal field forces, medical science liaisons, manufacturing teams, healthcare professionals (HCPs), and patients — each governed by different regulatory frameworks and content requirements.
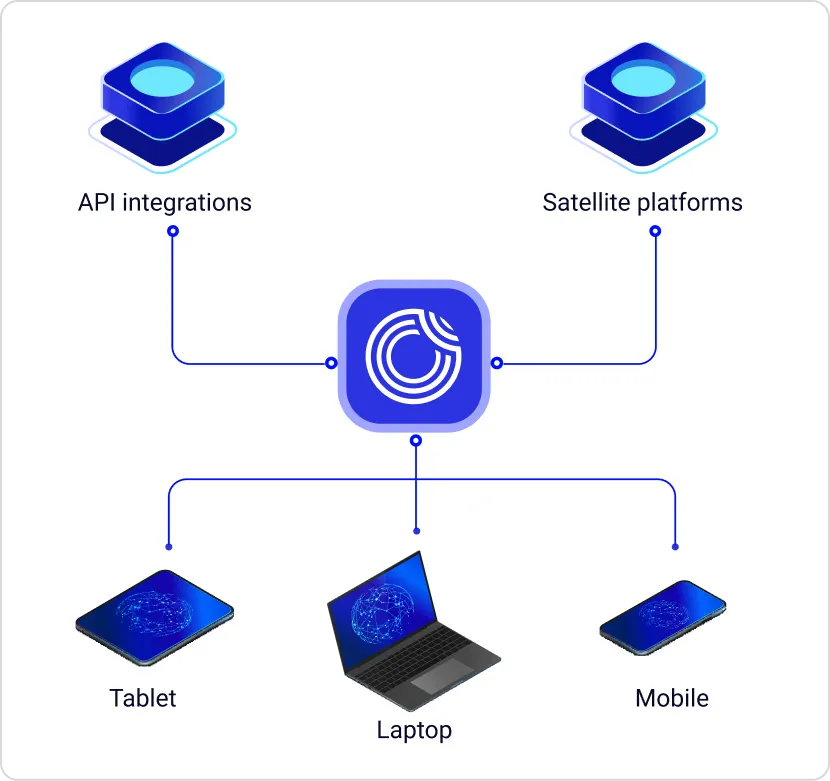
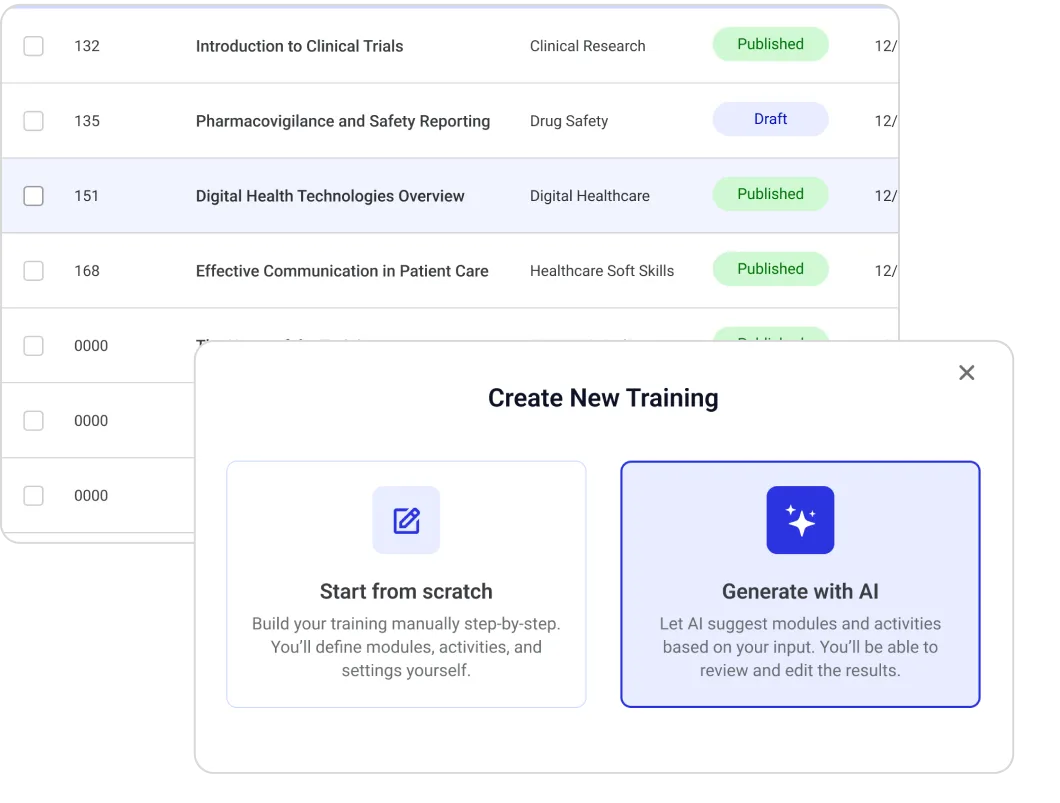
Opigno Enterprise is deployed by leading pharmaceutical organizations to manage this complexity from a single platform. Its multi-tenant, API-first, AI-powered architecture supports GxP-validated training environments, CME credit tracking, HCP portal integrations, and automated certification management — across any number of regions, and audience types.